Non-invasive in vivo metabolic profiling of inflammation in joints and entheses by optoacoustic imaging
Abstract Process
Aenean lacinia dui sed nisl porttitor, sed sodales quam euismod. Fusce et sem quam. Sed in sem quis est hendrerit vestibulum sed at risus. Ut volutpat ornare nulla, vitae ullamcorper sapien imperdiet non. Quisque tristique enim vel purus faucibus fringilla. Ut pellentesque arcu vel rhoncus convallis. Mauris tincidunt elementum dictum. Nunc luctus tortor mattis vestibulum lobortis. Praesent egestas, magna nec viverra feugiat, elit nisl dignissim nulla, non aliquam nisi sem vitae ipsum. In est magna, congue a ipsum sit amet, pulvinar sagittis augue. Nullam et purus egestas, vestibulum dolor eu, dignissim enim. Fusce pretium auctor urna, id volutpat erat tempor vitae. Ut et pharetra arcu, ornare sollicitudin ligula. Pellentesque vel facilisis risus. Mauris vestibulum tortor in quam hendrerit posuere. Ut viverra nibh neque, malesuada gravida orci semper et.
Duis sed purus in lacus pretium commodo. Mauris blandit mattis urna, id malesuada felis efficitur at. Cras tincidunt in urna id aliquet. Nunc ac imperdiet ex. Integer eget dictum enim. Nullam lobortis ut mauris at porttitor. Curabitur iaculis malesuada auctor.

Authors
Keywords
Arthritis; Enthesitis; Ultrasound; Imaging
Background
An in-depth metabolic characterization of joints and entheses at the tissue level can help in the early diagnosis and treatment selection for patients with inflammatory arthritis (1). However, current knowledge about the metabolic profiles of synovitis and enthesitis is limited. Multispectral optoacoustic tomography (MSOT), a novel metabolic imaging technology, could be used to undertake metabolic profiling of joints and entheses non-invasively using near-infrared multispectral laser to stimulate tissues and detect the emitted acoustic energy, enabling quantification of tissue components in vivo based on differential absorbance at multiple wavelengths (2, 3).
Objectives:
To explore the metabolic characteristics of arthritis and enthesitis using MSOT.
Methods:
Cross sectional study in healthy controls (HC), patients with rheumatoid arthritis (RA) and psoriatic arthritis (PsA). Participants underwent clinical, ultrasound (US), and MSOT examination of different anatomical regions (metacarpophalangeal joints, wrists, entheses of lateral epicondyles, patellar, quadriceps and Achilles tendons). MSOT-derived hemoglobin, oxygen saturation, collagen and lipid levels were measured. We calculated scaled mean differences (SMD) between affected and unaffected joints and entheses as defined by clinical examination or US using linear mixed effects models.
Results:
We obtained 1535 MSOT and 982 US scans from 87 participants (36 HC, 34 PsA, 17 RA) (Table 1). Entheseal tenderness was not associated with metabolic changes, whereas US enthesitis was associated with increased total hemoglobin, oxygen saturation and collagen content. In contrast, clinical and US arthritis showed increased hemoglobin levels but reduced oxygen saturation and reduced collagen content. Synovial hypertrophy was associated with increased articular lipids (Figure 1).
Conclusions:
MSOT allows non-invasive characterization of metabolic changes in arthritis and enthesitis. Our findings can be interpreted as a reflection of increased synovial cellularity, collagen degradation, and metabolic demand in synovitis, and of an increased tissue apposition and vascularization in enthesitis. Our results suggest that synovitis and enthesitis do not only differ at the clinical and anatomical-functional level, but also exhibit divergent metabolic changes.
References:
- Falconer J, et. al. Arthritis Rheumatol. 2018;70(7):984-99.
- Regensburger AP, et. al. Biomedicines. 2021;9(5).
- Regensburger AP, et al. Nature Medicine. 2019;25(12):1905-15.
Tables and Figures
Table 1: Baseline patient characteristics
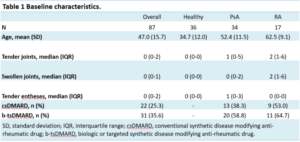
Figure 1: Scaled differences and 95% confidence intervals of MSOT-measured metabolite values by clinical and ultrasonographic findings of enthesitis (A-C) and arthritis (D-F).
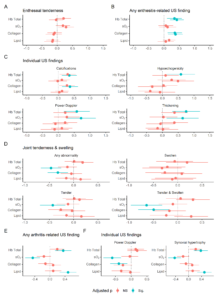
Enthesitis plots are depicted in A-C and arthritis plots in D-F. Two differences are plotted for each metabolite indicating two multispectral processing algorithms used for estimation. P values were adjusted for multiple testing using a false discovery rate of 5%. NS, not significant. sO2, oxygen saturation.